Hauptinhalt
The Lewis superacid Al[N(C6F5)2]3 and its higher homolog Ga[N(C6F5)2]3 – structural features, theoretical investigation and reactions of a metal amide with higher fluoride ion affinity than SbF5
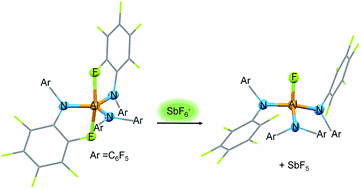
Herein we present the synthesis of the two Lewis acids Al[N(C6F5)2]3 (ALTA) and Ga[N(C6F5)2]3 (GATA) via salt elimination reactions. The metal complexes were characterized by NMR-spectroscopic methods and X-ray diffraction analysis revealing the stabilization of the highly Lewis acidic metal centers by secondary metal–fluorine contacts. The Lewis acidic properties of Al[N(C6F5)2]3 and Ga[N(C6F5)2]3 are demonstrated by reactions with Lewis bases resulting in the formation of metallates accompanied by crucial structural changes. The two metallates [Cs(Tol)3]+[FAl(N(C6F5)2)3]− and [AsPh4]+[ClGa(N(C6F5)2)3]− contain interesting weakly coordinating anions. The reaction of Al[N(C6F5)2]3 with trityl fluoride yielded [CPh3]+[FAl(N(C6F5)2)3]− which could find application in the activation of metallocene polymerization catalysts. The qualitative Lewis acidity of Al[N(C6F5)2]3 and Ga[N(C6F5)2]3 was investigated by means of competition experiments for chloride ions in solution. DFT calculations yielded fluoride ion affinities in the gas phase (FIA) of 555 kJ mol−1 for Al[N(C6F5)2]3 and 472 kJ mol−1 for Ga[N(C6F5)2]3. Thus, Al[N(C6F5)2]3 can be considered a Lewis superacid with a fluoride affinity higher than SbF5 (493 kJ mol−1) whereas the FIA of the corresponding gallium complex is slightly below the threshold to Lewis superacidity.
Veröffentlichung: The Lewis superacid Al[N(C6F5)2]3 and its higher homolog Ga[N(C6F5)2]3 – structural
features, theoretical investigation and reactions of a metal amide with higher fluoride ion affinity than SbF5.
J. F. Kögel, D. A. Sorokin, A. Khvorost, M. Scott, K. Harms, D. Himmel, I. Krossing, J. Sundermeyer, Chem. Sci. 2018, 9, 245–253. DOI: 10.1039/C7SC03988C