Main Content
Tissue separation and signal transduction by FGF receptors
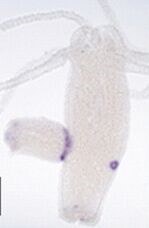
(Hassel and Bieller, 1996; Hassel et al., 1998; Sudhop et al., 2004; Hasse et al., 2014; Lange et al., 2014; Holz et al., 2017; Holz et al., 2020; Suryawanshi et al., 2020)
Cnidaria are one of the earliest branching metazoan phyla, and budding is their mode of asexual propagation. In a unique process of tissue separation, buds detach from the parent, when directly neighboring cells of the two monolayered epithelia separate. The fibroblast growth factor receptor (FGFRa), Kringelchen, controls bud detachment, and the second Hydra FGFRb is as well strongly expressed at the bud base (Suryawanshi et al., 2020). When Kringelchen is knocked down by either phosphorothioate antisense oligonucleotides, by the inhibitor SU5402 or by a dominant-negative transgene of FGFRa, detachment is prohibited (Sudhop et al 2004; Hasse et al 2014). Ectopic expression of Kringelchen, in contrast, induces ectopic tissue separation of the body column. The signaling pathway likely proceeds through one or two Rho A homologs, Rock and myosin II to target the actin cytoskeleton and to induce massive cell shape changes at the bud base (Holz et al. 2017; 2020). The function of the RhoA homologs is under investigation using an in situ approach for activated Rho detection.
Since lithium ions inhibit bud detachment as well and act as an agent to inhibit inositol phospholipid synthesis and therewith PI/PKC signaling (Hassel and Bieller, 1996; Hassel et al 1998), we investigated whether a link to PI phospholipid signaling exists. We established PI(4,5)P2 and PI(3,4,5)P3 sensor lines in cooperation with A. Klimovich and T. Bosch, Kiel. The sensors indicate a clear activation of PIP2 signaling at the late bud base.
At least four Hydra FGFs may serve as region-specific acting ligands (Lange et al. 2014). Their function as well as the functions of a second FGFR (FGFRb) and a soluble FGFRL1 protein are under investigation.