Main Content
Dissecting Key basic Mechanisms how Desmosomes control cellular Functions
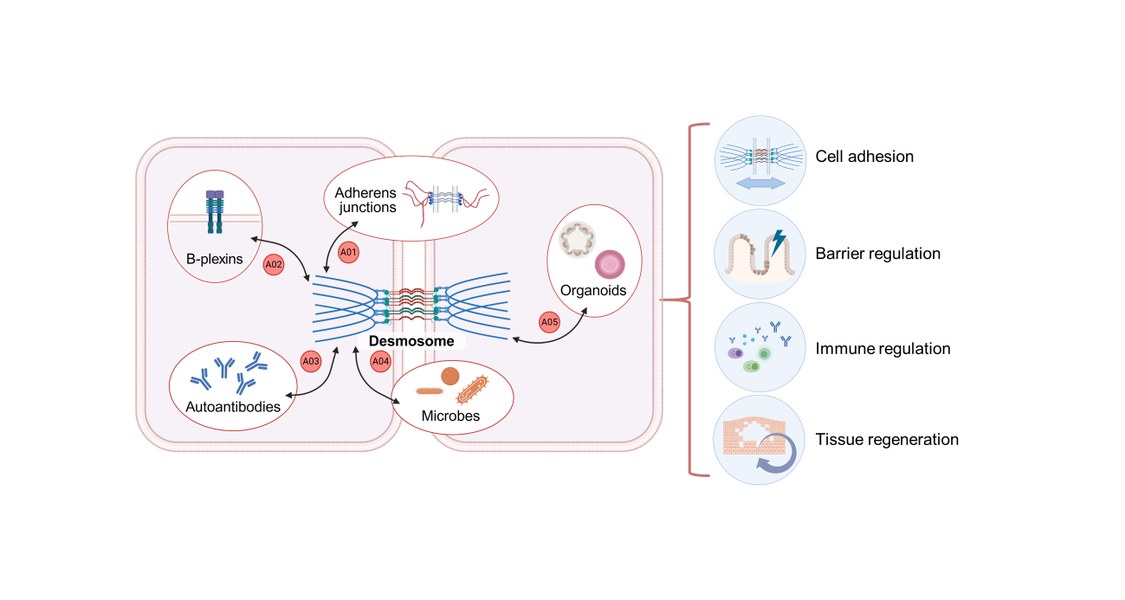
Inhalt ausklappen Inhalt einklappen A01 - Adherens junction and desmosome interactions in epithelial barriers in health and autoimmune disease
Carien Niessen, Department Cell Biology of the Skin and CECAD, University of Cologne
Carsten Grashoff, Institute of Integrative Cell Biology and Physiology, University of Münster
PhD students: Hanyin Zhang (Cologne), Valentina Pizza (Münster)
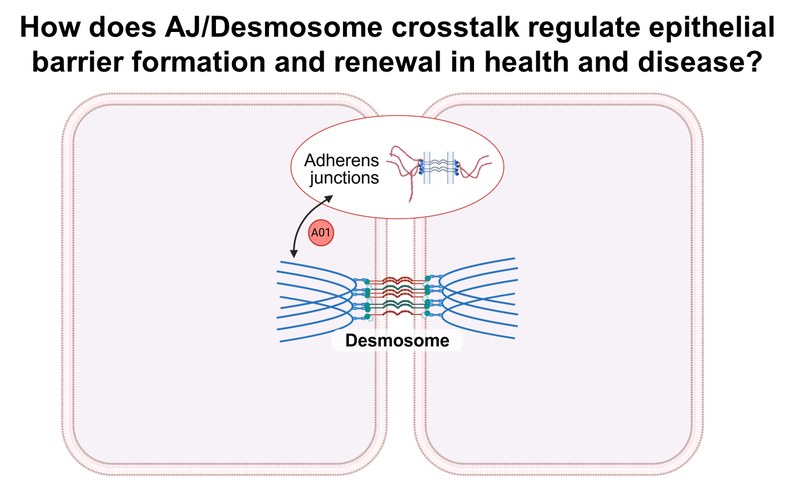
Project: A01 investigates how adherens junctions (AJs) and desmosomes (DSMs) coordinate their composition, stability, and turnover to regulate epithelial morphogenesis and regeneration in health and disease. Combining molecular force sensors and optomechanics with 2D, 3D, and in vivo models, we investigate the organization, mechanics, and function of AJs and DSMs at the molecular, cellular, and tissue levels in simple and stratified epithelia.Inhalt ausklappen Inhalt einklappen A02 - Control of desmosome function by B-plexins
Thomas Worzfeld, Institute of Pharmacology, UMR
Thomas Schmitt, Department of Anatomy, LMU Munich
PhD students: Yining Li (Marburg), Shahrzad Ameri (Munich)
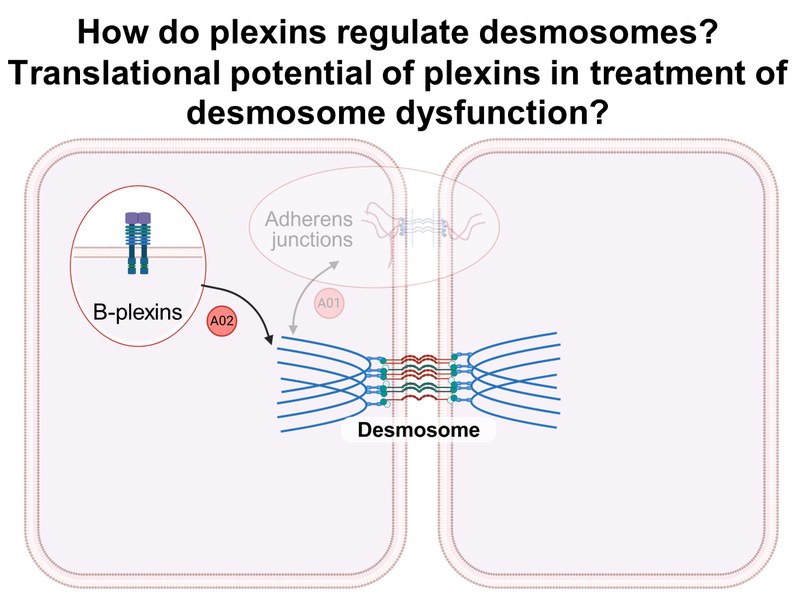
Project: A02 analyzes the B-plexin-mediated regulation of desmosome ultrastructure and function using biochemical and functional assays and confocal and high-resolution microscopy, as well as signaling events downstream of B-plexins that are important for desmosome localization and function. The pathophysiological role of B-plexins is investigated in preclinical mouse models of pemphigus vulgaris.Inhalt ausklappen Inhalt einklappen A03 - Super-resolution force spectroscopy to delineate early pathogenic effects of autoantibodies and cytokines in desmosome dysfunction
Michael Fuchs, Department of Anatomy, LMU Munich
PhD student: Fatemeh Lotfi
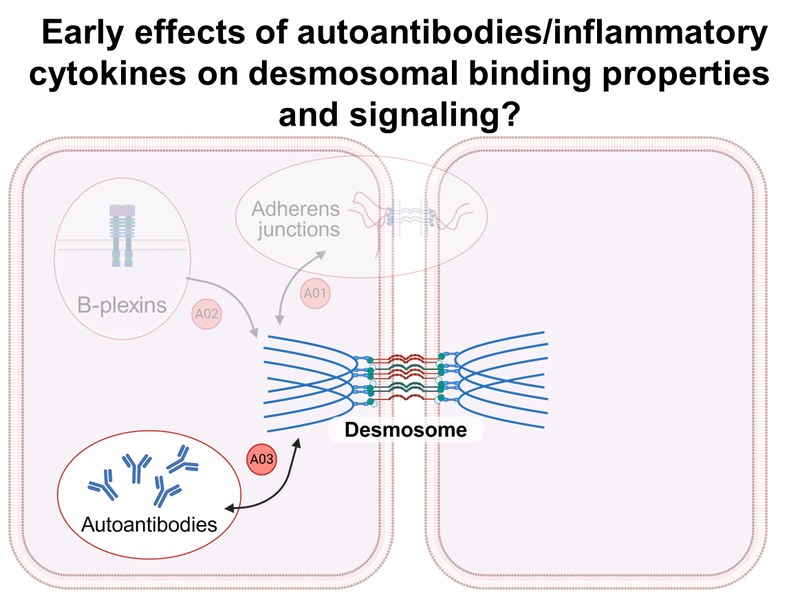
Project: A03 investigates the biophysical binding properties of desmosomal cadherins and the precise sequence of events following a pathogenic stimulus, i.e., autoantibodies or cytokines. For this purpose, atomic force microscopy (AFM), single molecule force spectroscopy (SMFS), and stimulated emission depletion (STED) microscopy are used in combination with biochemical approaches.
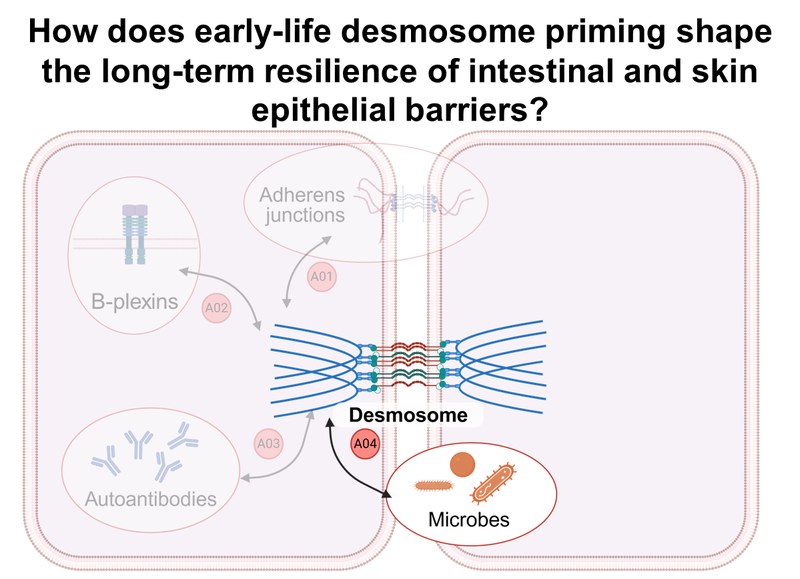
Inhalt ausklappen Inhalt einklappen A04 - Contribution of early life microbial priming to desmosome-mediated barrier function in cutaneous and intestinal epithelia under steady-state and inflammatory conditions
Mercedes Gomez de Agüero Tamargo, Institute for Systems Immunology, Max Planck Research Group, JMU Würzburg
Benjamin Misselwitz, Department of Internal Medicine II, LMU Munich
PhD students: Vaishnavi A. Menon (Würzburg), Ryan Scicluna (Munich)
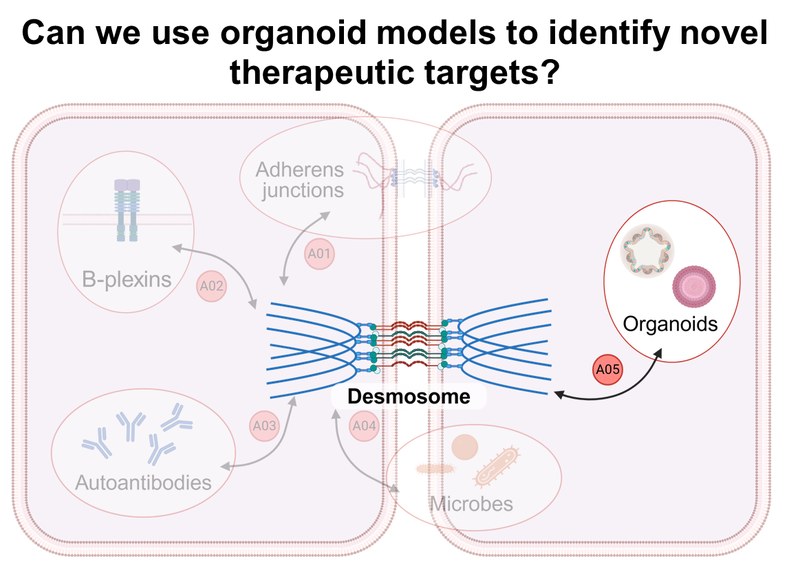
Project: A04 investigates the influence of prenatal microbial priming on desmosomal epithelial integrity in both healthy and inflamed states using organoids and murine imiquimod skin inflammation and colitis models. Human and murine epithelial cells from healthy and engineered skin and intestinal tissue will be used to generate 3D organoid models. Furthermore, targeted, microbiologically based interventions to restore damaged epithelial barriers will be developed.Inhalt ausklappen Inhalt einklappen A05 - Desmosome regulation in organoid models for mucosal and epidermal barrier dysfunction
Kai Kretzschmar, Mildred Scheel Early Career Centre for Cancer Research, UKW
Michael Meir, Department of General, Visceral, Transplant, Vascular and Pediatric Surgery, UKW
PhD students: Lena Paschmanns, Madhavi Hegde, Muthita Khamwong
Project: A05 develops organoid models of epidermal and mucosal desmosomal dysfunction using genetically modified mouse models. Tissue-specific phenotypes of mutant organoids will be comparatively investigated using electron microscopy, atomic force microscopy, 3D whole-body imaging, (single-cell) proteomics, and transcriptomics. Furthermore, we are establishing a biobank of organoids from healthy donors, IBD, and pemphigus patients.






