10.03.2026 When stress reprogrammes proteins
A Marburg research team around MiNu PI Gert Bange (Project 1) discovered a new molecular switching mechanism in bacteria.
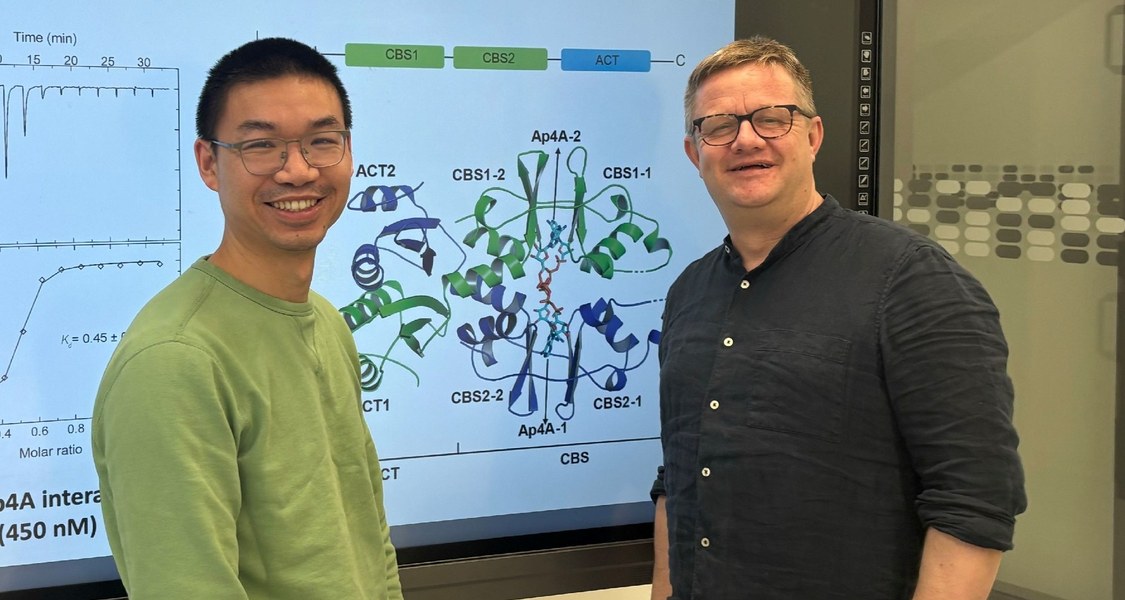
An international research team led by Prof. Dr Gert Bange from the Centre for Synthetic Microbiology at Philipps University of Marburg, who is also PI in RTG 2937, has uncovered a previously unknown mechanism by which bacteria adapt their cellular processes under stress. Together with Prof. Jade Wang (University of Wisconsin-Madison, USA), who is also Mercator Fellow of RTG 2937, the researchers demonstrate that the stress molecule AP4A specifically intervenes in the regulation of key metabolic processes. The findings have been published in the journal Nature Communications (DOI: https://doi.org/10.1038/s41467-026-70006-0).
Chemical tags control cell activity
Cells often regulate their proteins through a process known as acetylation – small chemical modifications that influence protein activity. To ensure this regulation remains flexible, such tags must be specifically applied and removed.
The Marburg team has now demonstrated that, under stress, the molecule AP4A acts like a switch. It stabilises the protein AcuB, which in turn inhibits a key enzyme that normally removes acetyl groups. As a result, numerous proteins remain more altered – the cell’s metabolism is readjusted.
Link between stress and metabolism
“For the first time, we have been able to understand in detail how AP4A binds to its target protein and thereby specifically influences protein acetylation,” says lead author Dr. Liujuan Zheng, a Chinese visiting researcher in Gert Bange’s laboratory. “Under stressful conditions, the molecule acts as an active signal – it shifts the balance of protein regulation and enables the cell to adapt rapidly.”
Lab head Gert Bange puts the findings into context: “This allows us to identify a new central switching point between stress signals and metabolism. Such mechanisms help us understand how cells coordinate the reorganisation of their internal processes under stress.”
Using modern structural and analytical methods, the team deciphered the molecular details of this regulatory mechanism.
Relevance for humans too
Particularly noteworthy: the bacterial enzyme studied resembles human histone deacetylases – key targets of modern cancer drugs. “The regulation of protein acetylation is widespread in evolutionary terms,” explains Bange. “As AP4A is also found in humans, comparable control mechanisms could exist. This opens up new perspectives for understanding metabolic regulation and stress biology.”
The study was supported by the German Research Foundation’s (DFG) Research Training Group 2937 ‘Microbial Nucleotide Metabolism’, the Max Planck Society, and the LOEWE Exploration Grant from the State of Hesse.
Original publication: Liujuan Zheng et al, Nature Communications (2026), https://www.nature.com/articles/s41467-026-70006-0
This article is a translation of the original German press release.