Main Content
Research
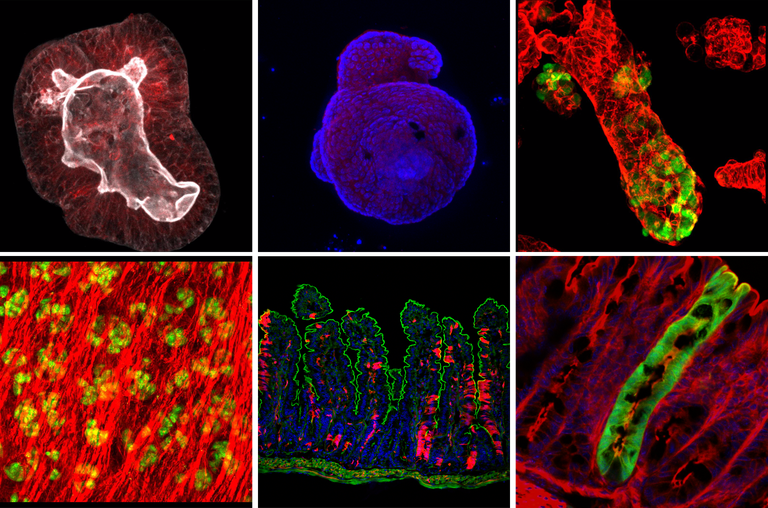
Research Focus: Western Lifestyle and Colorectal Cancer Risk
Colorectal cancer (CRCs) is not a disease of the elderly anymore and affects unfortunately more and more young adults. Already now every tenth CRC is diagnosed in persons below the age of 50 and it is predicted that the incidence of these “early-onset” CRCs will drastically rise in the next years. Strikingly, early-onset CRCs are often more aggressive and display very diverse clinicopathological and molecular characteristics, when compared to conventional CRCs found in older patients and it is hence believed that early-onset and late-onset CRC are distinct tumor-entities that follow different developmental routes. Our research team is dedicated to find out more about the causes for the steep increase of early-onset CRC and its adverse features.
In order to do so, we are focussing on one of the main environmental factors associated with early-onset CRC development, a “western” lifestyle. Especially adaption of western-style diet (WSD), has been described as a potent CRC risk factor and we suspect WSD to be a main driver behind the increase of early-onset CRCs. In several research lines, we are addressing how WSD predisposes to CRC and in the further course influences CRC development, progression and response to anti-cancer treatment.
Western-style diet as "creator" of a tumor promoting environment in the gut WSD has multiple, well-known profound effects on the gastrointestinal tract, which cumulatively increase the susceptibility to intestinal cancer, such as dysbiosis, intestinal barrier damage and low-grade intestinal inflammation. Hence, excessive consumption of WSD can predispose the individual to intestinal cancer by establishing an inflammatory and pro-tumorigenic environment in the gut. In this line of research we are analysing how WSD triggers dysbiosis and inflammation in the gut in order to identify measurable alterations that can be linked to increased CRC risk and help to identify persons at risk to develop CRC.
Western-style diet as "controller" of intestinal cellular identity We suspect that lifestyle factors, like WSD additionally increase CRC risk by disturbing intestinal stem cell homeostasis. We have previously shown that WSD, similar to inflammation, triggers the reprogramming of differentiated intestinal epithelial cells to stem-like cells, so-called “alternative stem cells, which in that way acquire the ability to serve as an additional pool of tumor-initiating cells. The expansion of cellular targets for tumor-initiating mutations might in turn, accelerate tumorigenesis. Hence, we are aiming on identifying transcriptional, epigenetic and metabolic alterations caused by WSD, in order to identify intracellular pathways and programs that can be targeted to restore perturbed intestinal stem cell homeostasis and minimize CRC risk.
Western-style diet as "influencer" of CRC development In this research line we are studying whether WSD directs CRC development towards specific tumor subtypes that are more frequently found in young CRC patients. By combining mouse CRC models with clinical data, we aim to reveal how WSD influences the development of specific histopathological, molecular, and immunological features of CRCs and moreover, whether WSD has effects on the tumors’ response to chemo- or immunotherapy.
However, in our lab it is not all about diet and CRC! We are currently expanding our research to other types of cancers and diseases associated with western-lifestyles and to other lifestyle factors associated with early-onset diseases.