Hauptinhalt
2019
Inhalt ausklappen Inhalt einklappen LANGMUIR: "Orientational and Crystalline Order of Copper Phthalocyanine Films on Gold: the Role of Substrate Roughness and Cleanliness"
Michael Kothe and Gregor Witte
Langmuir 35, 42, 13570-13577 (2019) • DOI: 10.1021/acs.langmuir.9b02658
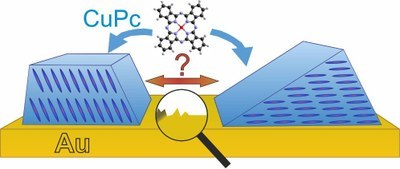
Although metal phthalocyanines are widely used in optoelectronic devices, e.g. as hole-transport and electron-blocking layers, or as UV-stable dyes, their multilayer growth on metal substrates has surprisingly not been studied very systematically. Even for CuPc, one of the most widely studied representatives of phthalocyanines, contradictory structures are reported for films grown on gold, a common electrode material, suggesting that the influence of actual substrate surface properties on film growth has not been sufficiently considered. In this study, we analyze the growth of CuPc films on gold substrates for thicknesses ranging from initial seed layer to thick multilayers (50 nm) by combining near-edge X-ray absorption spectroscopy (NEXAFS) with atomic force microscopy (AFM) and X-ray diffraction (XRD). To study the influence of surface roughness, we compare the formation of CuPc films on well-ordered Au(111) and sputter deposited polycrystalline gold substrates and also investigate the influence of surface contamination by exposing these gold surfaces to air before film growth. While on clean gold substrates, CuPc molecules exclusively adopt a recumbent orientation and form -oriented films, they also grow in an upright orientation on contaminated gold surfaces. On Au(111) this leads to a coexistence of - and (100)-oriented regions, whereas only (100)-oriented films are formed on contaminated polycrystalline gold. Remarkably, the -oriented films consist of extended but isolated crystalline islands, resulting in large overall roughness, whereas the (100)-oriented films consist of rather small domains, but have significantly lower film roughness.Inhalt ausklappen Inhalt einklappen ACS APPL. MATER. INTERFACES: "Controlling Interface Morphology and Layer Crystallinity in Organic Heterostructures: Microscopic View on C60 Island Formation on Pentacene Buffer Layers"
Andrea Huttner, Tobias Breuer and Gregor Witte
ACS Appl. Mater. Interfaces, 11, 38, 35177-35184 (2019) • DOI: 10.1021/acsami.9b09369
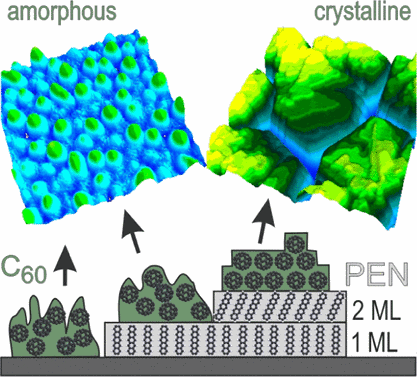
Controlling the crystallinity of organic thin films is an important aspect in the improvement of organic electronic devices. Howev-er, due to high molecular mass, structural anisotropy, and weak intermolecular van der Waals bonding, crystalline ordering is not easily accomplished. While film preparation at elevated substrate temperature often improves the crystalline quality, this ap-proach cannot be applied to temperature-sensitive materials such as plastic foils used as substrates for flexible electronics. Here, we examine in detail a low-temperature approach to improve film crystallinity by using ultra-thin pentacene (PEN) buffer layers that allow crystalline growth of Buckminsterfullerene (C60) thin films while without such buffer layers only amorphous fullerene films are formed upon room temperature deposition on various support substrates. Remarkably, this effect depends critically on the thickness of the PEN buffer and requires a thickness of at least two monolayers to induce crystalline growth whereas a buffer layer consisting of a monolayer of PEN again yields amorphous C60 films. Combining crystallographic investigations by X-ray dif-fraction and atomic force microscopy measurements we determine distinct nucleation sites on buffer layers of different thickness, which are correlated to the amorphous, respectively crystalline C60 islands. Our microscopic analysis reveals distinct differences for the nucleation and diffusivity of fullerenes on the PEN monolayer and on thicker buffer layers, which are attributed to the mo-lecular arrangement in the PEN monolayer. Finally, we show that the crystalline C60 films are exclusively (111)-oriented and the fullerene islands are even heteroepitaxially aligned on the PEN buffer.Inhalt ausklappen Inhalt einklappen NANOSCALE HORIZ.: "Growth of Extended DNTT Fibers on Metal Substrates by Suppression of Step-Induced Nucleation"
Maximilian Dreher, Dayeon Kang, Tobias Breuer and Gregor Witte
Nanoscale Horiz., Just Accepted (2019) • DOI: 10.1039/C9NH00422J
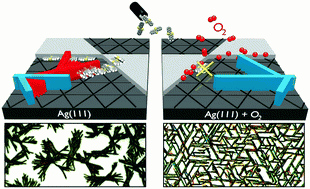
Due to their anisotropic optoelectronic properties, crystalline organic fibers constitute an interesting class of nanoscale materials with great potential for integration into future optoelectronic devices based on organic-inorganic hybrid systems. While chemical synthesis allows for flexible tailoring of electronic molecular properties, structural control of hybrid structures is hampered by the incompatibility of traditional structuring methods such as, e.g. lithography, with these molecular materials. Therefore, other approaches must be used. Here we examine the formation of crystalline fibers of dinaphthothienothiophene (DNTT), a recently synthesized chemically robust organic semiconductor with high charge carrier mobility, upon film growth on noble metal substrates. Based on a comparison of the film growth on a regularly stepped, vicinal surface, we show that substrate steps affect the azimuthal molecular orientation in the seed layer. In particular, they induce a fiber orientation which competes with that of fibers formed on flat terraces and thereby strongly limits the fiber dimensions. We demonstrate a strategy to suppress this parasitical step-induced fiber nucleation by first exposing Ag(111) surfaces to oxygen, or even briefly to ambient condition, which causes a selective saturation of the active step sites, while subsequent deposition of DNTT yields strongly enlarged fibers that are epitaxially aligned on the (111) surface.Inhalt ausklappen Inhalt einklappen ACS APPL. MATER. INTERFACES: "Multiplexed Read-out of Enzymatic Reactions by Means of Laterally Resolved Illumination of Quantum Dot Electrodes"
Shuang Zhao, Johannes Völkner, Marc Riedel, Gregor Witte, Zhao Yue, Fred Lisdat, Wolfgang J. Parak
ACS Appl. Mater Interfaces 11 (24), 21830 - 21839 (2019) • DOI: 10.1021/acsami.9b03990
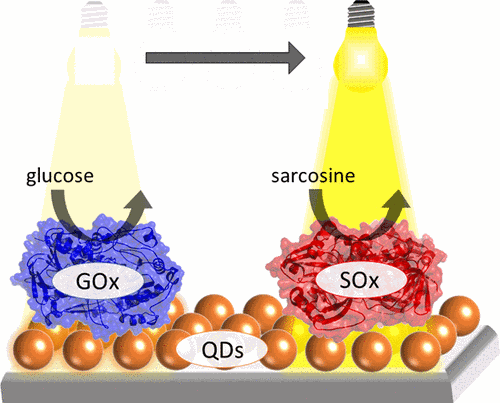
Triggering electrochemical reactions with light provides a powerful tool for the control of complex reaction schemes on photoactive electrodes. Here, we report on the light-directed, multiplexed detection of enzymatic substrates using a non-structured gold electrode modified with CdSe/ZnS quantum dots (QDs) and two enzymes, i.e. glucose oxidase (GOx) and sarcosine oxidase (SOx). While the QDs introduce visible light-sensitivity into the electrode architecture, GOx and SOx allow for a selective conversion of glucose and sarcosine, respectively. For the QD immobilization to the gold electrode a linker-assisted approach using trans-4,4’-stilbenedithiol (StDT) has been used, resulting in a generation of a photocurrent. Subsequently, GOx and SOx have been immobilized in spatially separated spots onto the QD electrode. For the local read-out of the QD electrode a new measurement set-up has been developed by moving a laser pointer across the surface to defined positions on the chip surface. The amplitudes of the photocurrents upon illumination of the GOx or SOx spot depend in a concentration-dependent manner on the presence of glucose and sarcosine, respectively. This measurement also allows for a selective detection in the presence of the other substance. The set-up demonstrates the feasibility of multiplexed measurements of enzymatic reactions using a focused light pointer resulting in an illumination area with a diameter of 0.3 mm for analyzing spots of different enzymes. Moving the laser pointer in x- and y-direction and simultaneously detecting the local photocurrent also allows a spatial imaging of enzyme immobilisation and activity distribution. Here not only the spot dimensions, but also the activity of the enzyme can be verified.Inhalt ausklappen Inhalt einklappen ADV. FUNCT. MATER.: "A Solvent-Free Solution: Vacuum-Deposited Organic Monolayers Modify Work Functions of Noble Metal Electrodes."
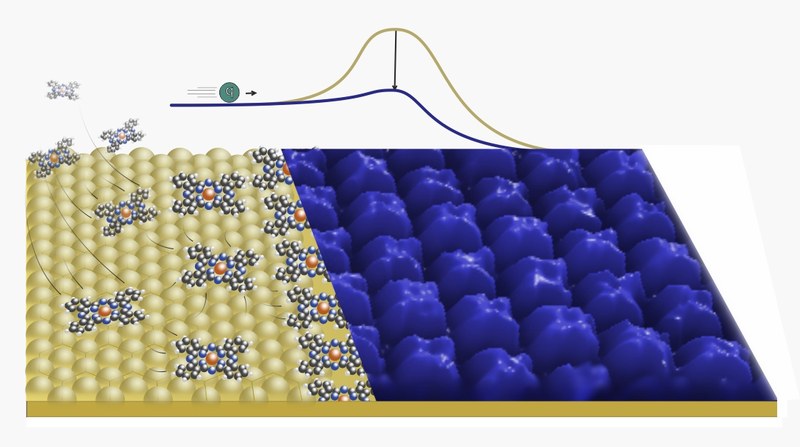
F. Widdascheck, A. A. Hauke, G. Witte
Adv. Funct. Mater., 1808388 (2019) • DOI: 10.1002/adfm.201808385 • see also press announcement and research outreach article
The energy level alignment between organic semiconductors (OSCs) and the respective (metal) electrodes in organic electronic devices is of key importance for efficient charge carrier injection. For many years, researchers have attempted to control this energy level alignment by means of functional self‐assembled monolayers or the insertion of thin injection layers (made, e.g., of doped OCSs or pure dopants). The present work demonstrates an alternative to these approaches, namely the use of phthalocyanine monolayers as contact primers, which are deposited onto noble metal electrodes by means of vacuum deposition. It is shown that polar as well as non‐polar phthalocyanines modify the work functions of clean Au(111) and Ag(111) surfaces as a function of their coverage and thus enable quantitative control of the metal work functions. This behavior is successfully replicated for the respective polycrystalline metal surfaces and it is found that full monolayers can even withstand air exposure when protected by sacrificial multilayers, which are afterward removed by thermal desorption.Inhalt ausklappen Inhalt einklappen J. PHYS. CHEM. C: "Titanylphthalocyanine Films on Ag(111): an Epitaxial Metal/Organic Hetero-System with Exceptional Smooth Surface."
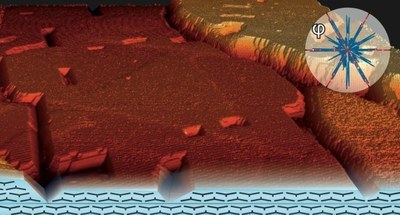
M. Kothe, F. Widdascheck, G. Witte
J. Phys. Chem. C 123, 6097 (2019) • DOI:10.1021/acs.jpcc.9b00506
The fabrication of smooth organic semiconductor films with homogeneous thickness is of key importance for the improvement of organic electronic devices and realization of well-defined molecular hetero-structures. While many pi-conjugated molecular materials form highly ordered monolayers on (single) crystalline metal substrates, further deposition typically obeys a Stranski-Krastanov growth and results in considerable layer roughness. Here, we examine the evolution of titanyl-phthalocyanine (TiOPc) films on Ag(111) for thicknesses ranging from initial seed layer to thick multilayers (200 nm) by combining scanning probe microscopy (STM, AFM) with X-ray diffraction (XRD) and synchrotron based photoelectron spectroscopy (XPS, NEXAFS). While the crystallinity of the TiOPc films increases with substrate temperature during growth, even at temperatures close to the onset of desorption, extended and molecularly flat islands are formed, that cover more than 80% of the substrate area resulting in exceptional smooth layers. The crystalline TiOPc films exclusively exhibit the Phase I polymorph where molecular planes are oriented nearly parallel to the substrate surface and adopt an alternating (up/down) stacking of their titanyl units forming stable bilayer units. Like the first bilayer also TiOPc multilayers are epitaxial aligned with respect to the substrate. STM and AFM data show further that the macroscopic film roughness is essentially due to rotational domain boundaries in the seed-layer which are not overgrown and result in characteristic macroscopic trenches. The high crystalline ordering and exceptional smoothness make this metal-organic hetero-system particularly suitable as molecular spacer layer and allows for thickness dependent studies of optoelectronic excitations and their dynamics.Inhalt ausklappen Inhalt einklappen J. PHYS. CONDENS. MATTER: "Charge transfer processes and carrier dynamics at the pentacene - C60 interface."
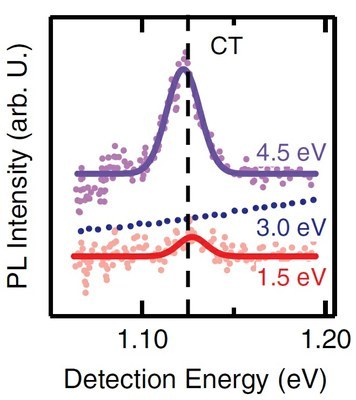
R. Döring, N. Rosemann, A. Huttner, T. Breuer, G. Witte, and S. Chatterjee
J. Phys.: Condens. Matter 31, 134001 (2019) • DOI: 10.1088/1361-648X/aafea7
Heterostructures of pentacene (PEN) and Buckminsterfullerene (C60) are frequently attracting scientific interest as a well-defined small-molecule model-system for the study of internal interfaces between two organic semiconductors. They are prototypical representatives forming a donor-acceptor combination for studies of fundamental optoelectronic processes in organic photovoltaics. Despite their importance in exciton dissociation, the energetics of their interfacial charge-transfer (CT) states and their microscopic excitation dynamics are not yet clarified and still being discussed. Here, we present steady-state and time-resolved photoluminescence measurements on stacked heterostructures between these two materials. All experiments are performed in the visible and near-infrared spectral regions as CT states are expected at energies below the fundamental electronic transitions of the respective bulk materials. A characteristic, interface specific emission at around 1.13-1.17 eV is found, which we attribute to an interfacial CT state. Its excitation-energy dependence reveals the intricate relaxation dynamics of excitons formed in both constituent materials. Moreover, the analysis of the dynamics of the C60 excitons shows that the lifetime of this state is reduced in the presence of an interface with PEN. This quenching is attributed to a long-range interaction, i.e., the relaxation of excitations into the interfacial CT state.Inhalt ausklappen Inhalt einklappen J. PHYS. CONDENS. MATTER: "Diels-Alder adduct formation at solid interfaces between Fullerenes and Acenes."
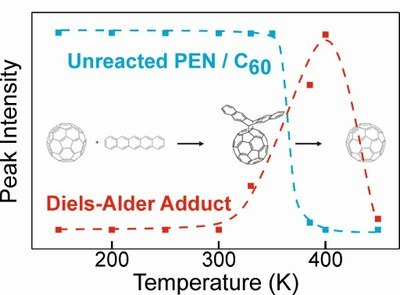
T. Breuer, T. Geiger, H. F. Bettinger, G. Witte
J. Phys.: Condens. Matter 31, 034003 (2019) • DOI: 10.1088/1361-648X/aaf01b
Understanding organic-organic interfaces is rather challenging due to their large complexity regarding morphology, molecular orientation at the interface, interdiffusion, and energetics. One additional important but often neglected aspect are chemical reactions occuring at such interfaces. For solid interfaces between pentacene and Buckminster-Fullerene (C60) recently very efficient Diels-Alder adduct formation has been reported. Considering the importance of pentacene/C60 as prototypical donor-acceptor combination to study fundamental processes in organic photovoltaics, understanding this effect is essential. In this work, we provide detailed NEXAFS-based investigations with respect to the temperature-dependence and reaction zone depth of this effect. Moreover, we widely vary the interface morphology and observe that the Diels-Alder adduct formation is most efficient for bulk heterojunctions of pentacene and C60. By also investigating further material combinations such as PEN/C60-PCBM and interfaces between C60 and functionalized acenes, we observe trends for the occurrence of the Diels-Alder adduct formation correlated with the different chemical properties of the involved compounds.